|
_SLIDE_1_
Phase equilibria Group Presentation
Critical point & applications
_SLIDE_2_
Explain the critical point and also the reason why critical points vary from material to material. Show some practical applications of the critical properties.
Group 2
Phase Equilibrium
_SLIDE_3_
Contents
What is the Critical Point?
The reason why critical point vary from material to material.
Practical applications.
_SLIDE_4_
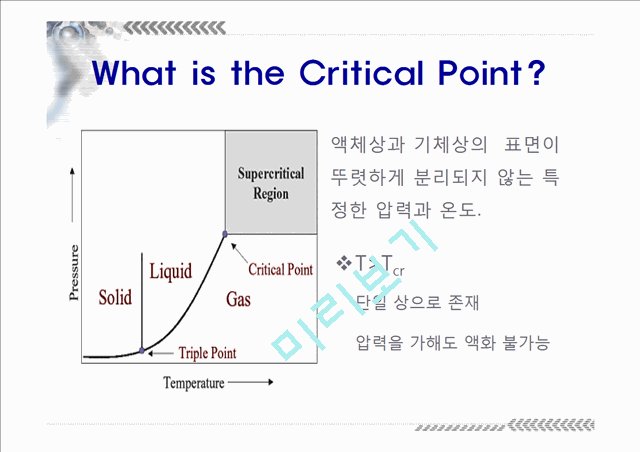
What is the Critical Point?
T>Tcr단일 상으로 존재압력을 가해도 액화 불가능
액체상과 기체상의 표면이 뚜렷하게 분리되지 않는 특정한 압력과 온도.
_SLIDE_5_
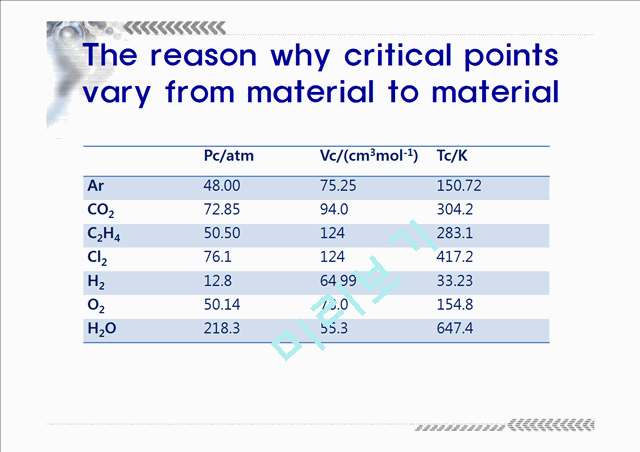
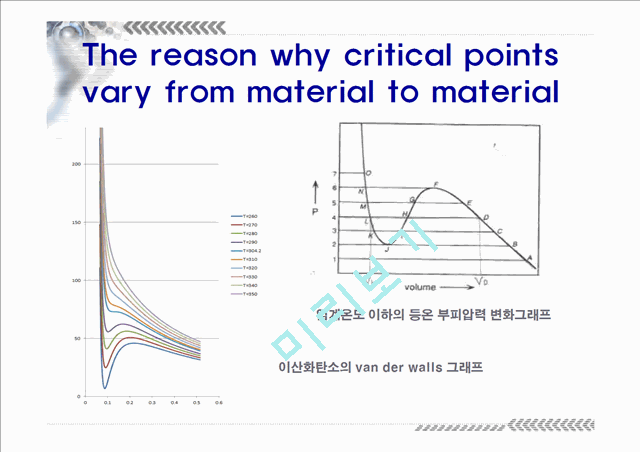
The reason why critical points vary from material to material
Pc/atm
Vc/(cm3mol-1)
Tc/K
Ar
48.00
75.25
150.72
CO2
72.85
94.0
304.2
C2H4
50.50
124
283.1
Cl2
76.1
124
417.2
H…(생략)
1.337
2.65
3.19
|
이상으로 적합한 초임계 유체상태로 변환
<2단계>
불순물을 초임계 이산화탄소에 용해시킨 후 분리조로 이동, 압력을 낮춰 서로 분리
<3단계>
분리된 기체 이산화탄소를 냉각하여 액체상태로 전환시킨 후 사이클 다시 반복
_SLIDE_14_
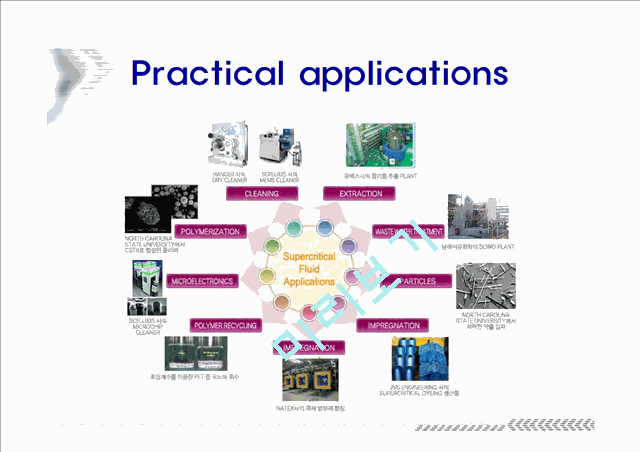
Practical applications
초임계 유체 건식 세정
- 반도체 웨이퍼 표면세정, 미세한 기공의 소재의 세정에 적용 가능
- 건조단계 불필요, 대상물의 손상방지, 경제성, 환경친화적인 장점을 갖음
<초임계 유체 건식세정 장치도>
_SLIDE_15_
Practical applications
Supercritical water oxidation technology
물의 초임계유체
유기물과 산소의 용해도 크게 증가
난분해성 유기물과 산소의 반응 증가
매우 빠르며, 100%에 가깝게 반응
폐수처리에 이용
_SLIDE_16_
References
김재덕 외 4명, 초임계 유체를 이용한 환경친화적 화학반응공정 개발 연구, 과학기술부, 2004
David R. Gaskell, 재료열역학, 홍릉과학출판사, 2009
Peter Atkins, Physical chemistry, Oxford, 2006
Principles of phase diagrams in material systems - P.Gordon, 1968
http://sfrl.kist.re.kr/
http://www.epartsmall.net/
http://www.reseat.re.kr/INDEX.jsp
http://ko.wikipedia.org
http://blog.naver.com/xuzte73hi
_NOTES_1_
2